News

Zhang Group: Efficient Nanocrystal Surface Cleaning by N-Heterocyclic Carbene
Efficient Nanocrystal Surface Cleaning by N-Heterocyclic Carbenes
By Emma Cook, UVA ChemSciComm
Perrin Godbold, Grayson Johnson, Akachukwu D. Obi, Rebecca Brown, Sooyeon Hwang, Robert J. Gilliard Jr., and Sen Zhang J. Am. Chem. Soc. 2021, 143, 2644-2648
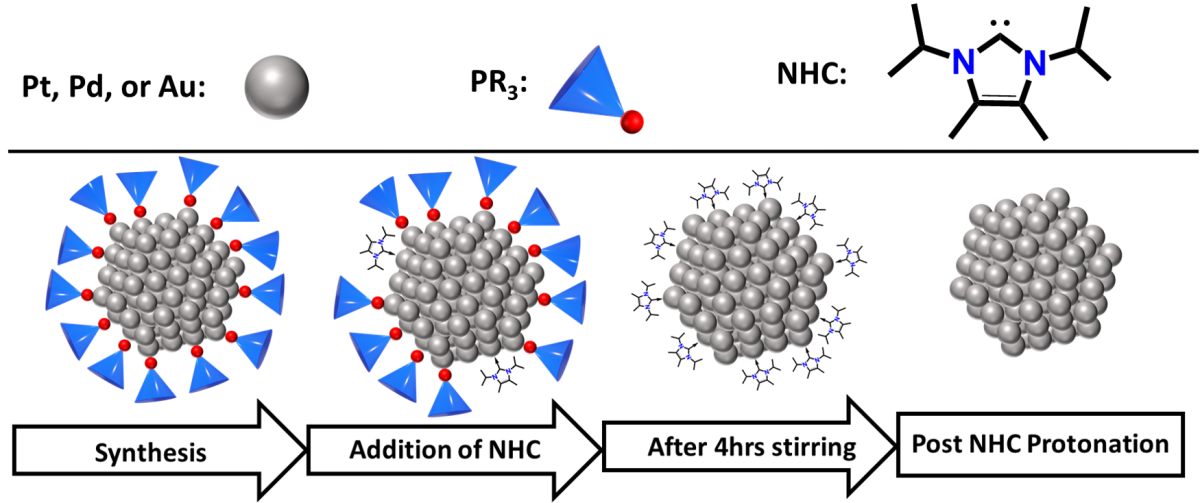
Nanocrystals (NC) are nanometer-scale particles made of metal atoms that play key roles in catalysis, medicine, and technology applications because of their unique structural and chemical properties, which can differ significantly from the bulk material. The surfaces of NCs are stabilized by surfactants, which are typically long carbon-hydrogen atom chains attached to a polar group that binds to the NC surface. These surfactants play critical roles in NC synthesis and are used to control NC shape, size, and growth rate. Despite their key role in synthesis, surfactant bulkiness can inhibit catalysis by NC because they block potential surface-active sites. Consequently, there have been many surfactant removal methods developed in order to make these surface-active sites accessible, but these methods are often insufficient for strongly bound surfactants, such as phosphines.
In collaboration, Drs. Sen Zhang and Robert Gilliard and coworkers have developed a surfactant removal method that utilizes N-heterocyclic carbenes (NHC). In this two-step process, trioctylphosphine (TOP) surfactants on platinum, palladium, and gold NC surfaces were first exchanged with NHC ligands. Then, surface bound NHCs were reacted with concentrated acid and were subsequently removed from the NC surface to expose the NC active sites. These surfactant-free NCs demonstrated improved catalysis toward reactions that are important to the development of alternative energy applications. This new strategy of surfactant removal to expose NC surface active sites will improve the ability for NCs to be used in not only catalysis but other relevant applications, such as alternative energy and biomedical sensing technologies.