News

Gunnoe Group: Stilbenes- Important Molecules in Need of a Synthesis Makeover
By Josh Prindle, UVA ChemSciComm
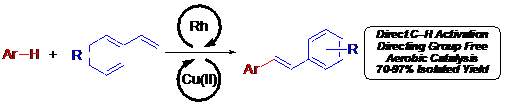
Article Link: Synthesis of Stilbenes by Rhodium-Catalyzed Aerobic Alkenylation of Arenes via C–H Activation
While stilbenes are found in nature as secondary plant metabolites, man-made stilbene derivatives find utility as precursors for the development of cancer-fighting drugs. The light-emitting properties of other stilbene derivatives make them excellent components in dyes, liquid crystal displays, and light-emitting diodes (LEDs). Current industrial methods for their synthesis require multiple steps and generate significant waste. There is therefore a need for new synthetic routes to these important commodity chemicals to satisfy demand and diminish the environmental footprint.
Dr. Brent Gunnoe and co-workers at the University of Virginia have recently developed a one-pot approach to synthesizing stilbene derivatives from abundant aryl and vinyl arene co-substrates. In the presence of a rhodium-based catalyst, which can selectively activate carbon-hydrogen bonds in the aryl co-substrate, stilbene products are selectively generated using an air-recycled Cu oxidant. The use of the rhodium catalyst is key to the improved reaction conditions, as it removes the need to prefunctionalize substrates common to other approaches, decreasing the amount of waste and improving the atom economy of the reaction.
By avoiding the laborious and wasteful task of separating and purifying precursor molecules and performing the entire synthesis in just one reactor, this approach simplifies the process of synthesizing stilbene derivatives. Dr. Gunnoe believes this new one-pot reaction offers the possibility for more efficient large-scale production of commercially relevant stilbenes. “We believe that the newly reported catalytic route developed by our group provides a substantial improvement in efficiency and can serve to significantly reduce chemical waste and energy consumption compared to existing synthetic methods. Thus, we anticipate that the catalytic process will be used for the preparation of existing and new compounds and materials that contain the stilbene unit.”